Introduction
The generic drug market in the United States is a critical component of the healthcare system, providing cost-effective alternatives to brand-name medications and contributing to significant savings for both patients and the healthcare system. The Inflation Reduction Act (IRA)1 of 2022 introduces several provisions aimed at reducing drug prices and redesigning the benefit to reduce out of pocket costs for some beneficiaries. While the full consequences of the IRA are not yet realized, these provisions have the potential to change how manufacturers of generic drugs find success moving forward.
Historical Perspective
The US Congress enacted The Drug Price Competition and Patent Term Restoration Act, better known as the Hatch-Waxman Act, in 1984 to balance incentives for innovation while fostering a robust generic drug market. The established framework did this in part by creating a streamlined process for generic drug approvals. Prior to passage of Hatch-Waxman, just 19%2 of prescriptions in the US were filled with generics and only 35%2 of top-selling pharmaceuticals had generic competitors after their patents expired. Moreover, it took 3 to 5 years for generics to enter the market after the patents on brand-name pharmaceuticals expired.
Current Generic US Landscape
Today, generics account for approximately 90%3 of all prescriptions dispensed, although they represent a lower percentage (12%)4 of total drug spending due to their lower prices. Moreover, the average effective patent life for small molecule drugs before generics enter the market today is 13 to 14 years5—demonstrating this balanced framework has proven successful in encouraging introduction of innovative drugs while ensuring swift market entry of generics. This success is also reflected in the savings generics drive in the health care system. The US Food and Drug Administration (FDA) estimates that new generic drugs approved in 2018, 2019, and 2020 resulted in approximately $53.3 billion in savings in each of those years.6 But importantly, these savings accumulate over time as more generics are approved providing long term value to the healthcare system while new brand drugs continue to enter the market. Overall, generics are a crucial tool in controlling costs and increasing access to drugs.
Generic drug manufacturers are experiencing increasing demand but also increasing pricing pressure from continued consolidation of buyers (e.g., wholesale buying consortia, pharmacy benefit managers [PBMs] and group purchasing organizations [GPOs]) that gain leverage to lower prices. They also face increasing competition from new companies within the US and abroad, further decreasing prices. Most alarmingly, as prices fall over time with increased competition, manufacturers are deciding to stop the manufacture of generics, particularly sterile injectables,7 as they have become unprofitable. This can lead to insufficient market supply and drug shortages, ultimately impacting the patients who need them the most.8
Generic manufacturers consider various factors prior to entering the market including market need, timing of regulatory submissions to ensure alignment with brand drug patent expirations, and the size of the financial opportunity (i.e., projected drug price and market share). Being the first generic to market under Hatch Waxman can provide the opportunity for a 180-day period of exclusivity9 during which time there is limited or no competition from other generics. In this way, the framework encourages generics to enter the market swiftly in order to capture market share before additional generics reach the market. When a single generic is on the market, the generic average manufacturer price (AMP) falls 39%. When two generics are on the market, the generic AMP falls to 54%. Generic prices continue to fall as the number of generic products on the market increase. When six or more generics enter the market, the generic AMP falls by a staggering 95%.10
Generic manufacturers are attracted to branded drugs with high-volume sales and larger financial opportunity prior to patent expiration where there is a stronger likelihood of achieving significant market share. Unfortunately, due to possible unintended consequences of provisions in the IRA, there may be a significant disruption to these historic dynamics, creating a new – and less beneficial – paradigm for generic manufacturers.
Components of the IRA
The provisions of the IRA most impactful to the drug industry are the lowering of costs for prescription drugs for Medicare beneficiaries and the government. The main components of the IRA that allow the Centers for Medicare & Medicaid Services (CMS) to execute on these objectives are:11
-
- CMS Drug Price Negotiation: CMS is authorized to negotiate maximum fair prices (MFPs) for certain high-expenditure, single-source drugs, and biological products covered by Medicare. The process targets drugs without a marketed generic or biosimilar competitor at the time of selection. Under certain circumstances, in the event there is an imminent biosimilar, the law allows for a pause in the process.
-
- Part D Redesign: Changes to the Part D benefit that will occur over 2 years beginning in 2024 and be fully implemented for the 2025 plan year. Changes include removing the enrollee 5% financial responsibility in the catastrophic phase, capping annual out-of-pocket (OOP) costs for beneficiaries for brand drugs at $2,000 annually, and eliminating the coverage gap by shifting more financial responsibility from Medicare to insurance plans and manufacturers for brand drugs. Products subject to MFPs will be exempt from manufacturer financial responsibility in the Part D benefit.
Impact of the IRA on Generic Manufacturers
Lower brand drug prices
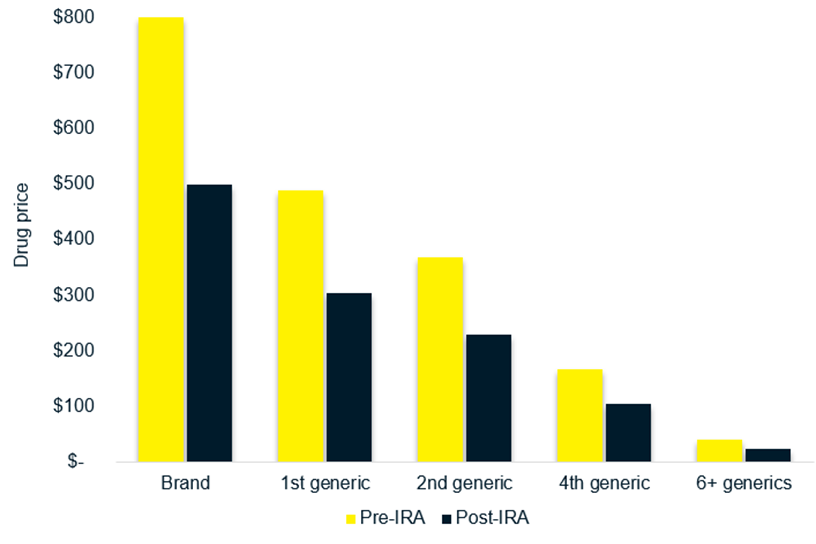
CMS will negotiate brand drug pricing with manufacturers for selected drugs and assign an MFP. The MFP set by CMS may reduce the price the generic manufacturer is able to set for the generic version of the drug. Reduced price differentials can lead to lower revenue for generic manufacturers, potentially deterring investment in manufacturing generic products (Figure 1). If fewer generic products enter the market, there will be less competition and the full cost savings potential of generics may not be realized.
Figure 1. Potential impact of CMS negotiated MFP on generic prices.10
Fewer brand indications
Under the IRA there may be less incentive for brand manufacturers to invest in additional indications for FDA-approved drugs. CMS now has the ability to negotiate drug prices for small molecules that have been on the market for 7 or more years, which is far earlier than the 13-14 years small molecules average before facing generic competition. This limits the time to recoup investment cost for new indications. In reaction to the loss of revenue in the years post MFP negotiation, brand manufacturers are likely to scale back clinical development programs since patent protections or regulatory exclusivities for new indications will not delay CMS negotiations.12 In disease areas where post-approval indications are common in bringing new treatment options, including cancer, chronic diseases and rare diseases, these impacts are expected to be more acute.
A decrease in new indications will reduce market share of branded products, making it financially challenging to develop and launch new generics. If fewer generics enter the market, price reductions may be limited.
Reduced investment in small molecules
CMS drug price negotiation reduces incentives for brand manufacturers to invest in small molecules due to the reduced value of patents. Small molecule drugs can be selected for negotiation 7 years post FDA approval, whereas biologics are afforded a longer runway on the market (11 years) before becoming eligible for negotiation. To navigate price pressures, brand manufacturers may shift research and development (R&D) to developing biologic drugs for complex conditions with higher price points and less competition, offering a longer time to achieve returns on investment. Pharmaceutical manufacturers have been shifting development away from small molecules toward biologics for quite some time, and this trend may intensify under the IRA.13
As a result, fewer small molecule drugs may be developed over time, leading to fewer generics entering the market. Small molecule medicines provide critical clinical benefits that cannot be replicated by biological medicines, such as the ability to reach therapeutic targets inside cells and cross the blood-brain barrier. They also provide great flexibility and convenience for patients as most can be easily self-administered, reducing barriers to treatment adherence.
Greater competition among generic manufacturers may also lead to further consolidation within the industry. Ultimately, fewer generic companies chasing fewer generic development opportunities will erode the current cost savings realized from the industry today.
Market access
Currently, when a new generic comes to market, it takes approximately 3 years before it is covered on over 50% of Medicare plans.3
Despite the assumption that CMS drug price negotiation will lower brand drug prices, the IRA as it is currently written does not restrict payers from preferring highly rebated high-cost brands over lower cost brands and generics. In fact, payers have publicly stated they expect brand manufacturers to continue to offer rebates to maintain formulary preference over new to market generics. They will continue to be incentivized to prefer high-cost drugs that offer rebates over lower cost products, further reducing brand net prices.14 Payers may place generics and biosimilars on higher tiers with higher copayments, making them more expensive for patients. Government investigators have noted PBMs and health plans may be incentivized to cover reference brand products that deliver rebates and support payers’ profits over lower cost generics, which may contribute to this dynamic. In 2022, the US Federal Trade Commission (FTC) stated that “rebates and fees may shift costs and misalign incentives in a way that ultimately increases patients’ costs and stifles competition from lower-cost drugs, especially when generics and biosimilars are excluded or disfavored on formularies.”15
Spillover effects
The impact of IRA drug negotiations will not be limited to MFP-negotiated drugs. A lower price on the selected drug will incentivize payers to negotiate lower prices for all related products in the same or similar therapeutic class. In addition, it is common for payers to align their commercial and Part D formularies. Payers have indicated that they will use published MFPs as a baseline for negotiations across their commercial business as well as Medicare to offset increased liabilities under Part D redesign.16
The IRA will have far reaching effects on generics, both in development and pricing. All branded drugs in the same class as an MFP drug will have increased pressures on price in both commercial and Medicare markets. New generics within these therapeutic classes will need to launch at prices low enough to incentivize payers to cover them while still maintaining profitability, which may prove to be an impossible task given the market trends created by the IRA.
Unintended Consequences
The IRA will fundamentally change the nature of the opportunity for new generics. Today’s functioning generic market delivers significant cost savings often by targeting the most successful branded products, the same products that will be targeted by the IRA. This could impact the extent to which generic manufacturers remain a critical driver of value to the health care system.
Unlock your PDF download
"*" indicates required fields



